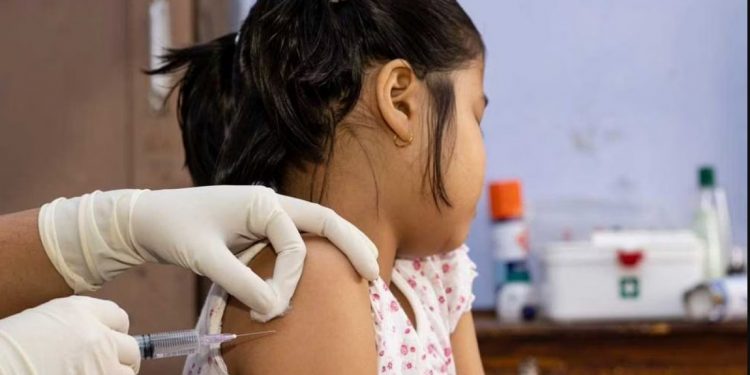
New Delhi: India’s drug regulator, the Drugs Controller General of India (DCGI) has granted restricted emergency use authorisation (EUA) for Biological E’s COVID-19 vaccine Corbevax for those aged five to 12 years. It has also given approval to Bharat Biotech’s Covaxin for children in the age group of six to 12 years. This information was given by Union Health Minister Mansukh Mandaviya here Tuesday.
The approval by DCGI for Corbevax and Covaxin for inoculating children below 12 years paves the way for giving protection to a majority of school going children. The decision assumes significance ahead of Prime Minister Narendra Modi’s meeting Wednesday with chief ministers on the emerging Covid-19 situation in India.
The DCGI has also granted EUA to Cadila for its ZyCoV-D for an additional dose of 3mg with a two-jab inoculation schedule 28 days apart for those aged above 12 years. ZyCoV-D is, currently, approved for a 2mg three-dose vaccination schedule.
Also read: Kids can register on CoWIN for vaccination with student ID cards
The DCGI approval comes following recommendations by the Subject Expert Committee (SEC) on Covid-19 of the Central Drugs Standard Control Organisation(CDSCO).
The SEC had last week reviewed the application of Biological E and Bharat Biotech seeking emergency use authorisation for Corbevax and Covaxin for use in children of five to 12 years and six to 12 years age group, respectively. It also reviewed Cadila’s application.
India began inoculating children aged 12-14 years on March 16. So far, more than 2.70 crore adolescents have been administered with the first dose of Covid-19 vaccine.